Helium atoms within tiny drops show an extremely fast energy exchange
Marcel Mudrich and E. Fasshauer of IFA are co-writers in a recent paper in the web periodical Physical Review X.
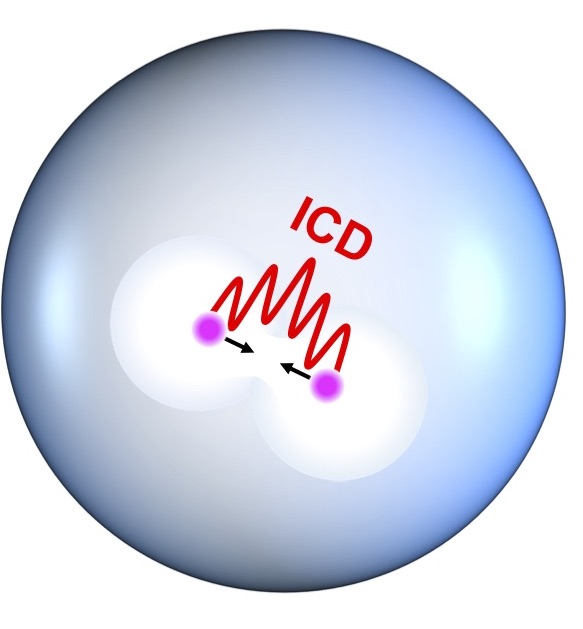
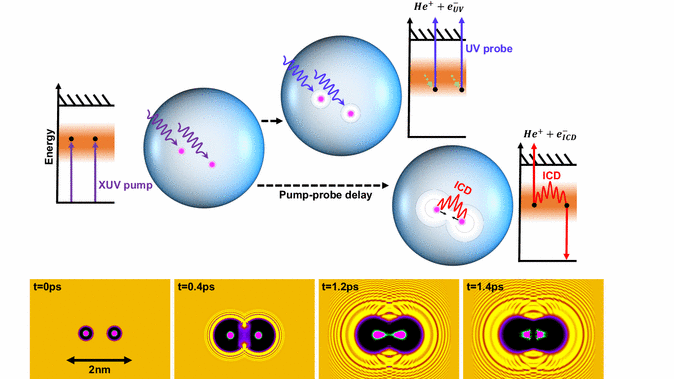
An isolated excited atom takes a relatively long time to get rid of its exitational energy, but in clusters of atoms or molecules a long list of interactions are able to further the relaxation processes considerably. One of the fastest processes is ICD - Interatomic Coulomb Decay. In this paper an international group of researchers has been studying the decay times in nanodrops of helium, and surprisingly fast decays occur; in the range of 400 femtoseconds.
In the experiments the times of decay have been measured by exiting the helium drops with a free electron extreme UV laser at many different intensities and drop sizes.
In the blue bubbles of helium single atoms are exited, and shortly after they bump together and exchange charge. In the yellow rectangles the dimensions and temporal frames are shown. Illustration from the paper. Click on the illustration to see an animation of the bubble dynamics.
Quantum mechanical calculations support a theoretical assumption of two excited atoms forming an empty bubble around each atom. Subsequently the bubbles then melt together, pushing the atoms towards each other, promoting the ICD-process.
Understanding the formation of bubbles in a relatively simple system like superfluid helium will help understanding of energy and chargeexchanges e.g. in proteines in biological systems or the effect of impurities in metals.
The paper, titled " Ultrafast resonant interatomic Coulombic decay induced by quantum fluid dynamics" can be found here.
A short resume from Physical Review X can be found here, and a longer press release is here.