In physics our main interests are related to dosimetry where we try to model and understand the detailed energy deposition of protons and heavier ions in the body as well as in detectors. Biology research is conducted by the Department of Experinental Clinical Oncology and is mostly concerned with radiobiology, that is measuring cell survival in proton, antiproton and carbon ion beams in vitro and in vivo. On the clinical side, we are intending to bridge the gap between basic and clinical research by developing cause-driven treatment planning and performing randomized phase III trials to prove the evidence for particle therapy.
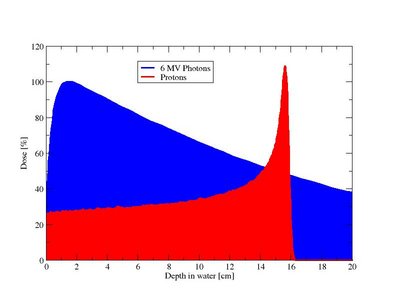
Radiotherapy with fast charged particles such as protons and heavier ions for cancer treatment is becoming more readily available due to several new facilities emerging around the world. Currently, particle therapy facilities with protons and heavier ions are available at more than 35 operational centers. Until January 2009, in total around 60.000 patients were treated with protons and 7000 with ions heavier than protons. Due to the inverse dose profile of the charged particles, a more conformal radiotherapy can be achieved as compared to conventional radiotherapy with photons. Thereby, the probability of achieving tumor control is effectively increasing while simultaneously reducing the risk of normal tissue complication [1–5].
Particle therapy, especially with ions heavier than protons, poses new scientific challenges for the understanding of the dosimetry and radiobiology. Whereas protons have radiobiological properties similar to what is known from photons, carbon ions induce more biological damage for the same physical dose. These effects are highly non-linear and they depend on several parameters, such as particle type and energy, tissue type, applied particle fluence (dose), etc. Given the complexity of cell biology, it is practically impossible to realize an exact model of radiation damage. Instead phenomenological approaches are used which have had some success in describing the radiobiological properties of particle radiation. So far, the Local Effect Model (LEM) [6–9] is one of the most adequate models describing the radiobiology. It is a derivative from track structure theory [10]. The LEM was applied for calculating the biological effective dose for carbon ion treatment at the Gesellschaft für Schwerionenforschung (GSI) Darmstadt, Germany, and is also used at the Heidelberg Ion-Beam Therapy Center (HIT), Germany, which started treating patients in November 2009.
Back to APTG main page